Morgan Milton, PhD
 Assistant Professor
Assistant Professor
Brody 5W-56
The Brody School of Medicine at East Carolina University
Greenville, NC 27834
phone: 252-744-1707
email: miltonm19@ecu.edu
Education
- B.S., Georgia Institute of Technology, Biology, 2009
- M.S., Georgia Institute of Technology, Biology, 2010
- Ph.D., Iowa State University, Biochemistry, 2016
- Post-doctorate, North Carolina State University
- Post-doctorate, RTI International
- Post-doctorate, East Carolina University
Research Interests
- Molecular mechanisms governing bacterial biofilm formation
- Signaling networks connecting quorum sensing and biofilm formation
- Antibiotic tolerance and resistance
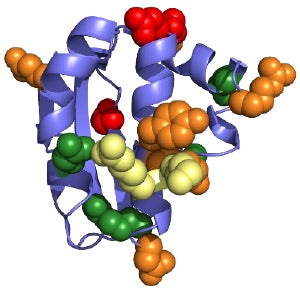 Biofilms allow bacteria to be up to 1,000-fold more resistant to antibiotics and the host immune response than free-swimming bacteria. Since biofilms are not required for bacterial viability, therapeutic interventions targeting biofilm formation are less likely to induce the undesired outcomes of bacterial resistance. Therefore, understanding how bacteria regulate biofilm formation would allow us to identify targets for next generation antimicrobial therapies. My research explores how non-Cholera Vibrio (NCV) bacteria regulate the formation of biofilms. NCV species are a significant threat to human health through food poisoning and wound infection, causing an estimated 80,000 illnesses per year in the United States. As ocean temperatures continue to rise due to climate change, Vibrio infections are expected to sharply increase. Vibrio species employ highly complex systems to regulate the formation of robust biofilms that are relevant to host infection. My goal is to uncover the molecular mechanisms of critical proteins in these pathways, using a wide range of biochemical, biophysical, and microbiological approaches. This work will answer many basic science questions about biofilm regulatory mechanisms and will allow us to identify potential intervention points for drug targets.
Biofilms allow bacteria to be up to 1,000-fold more resistant to antibiotics and the host immune response than free-swimming bacteria. Since biofilms are not required for bacterial viability, therapeutic interventions targeting biofilm formation are less likely to induce the undesired outcomes of bacterial resistance. Therefore, understanding how bacteria regulate biofilm formation would allow us to identify targets for next generation antimicrobial therapies. My research explores how non-Cholera Vibrio (NCV) bacteria regulate the formation of biofilms. NCV species are a significant threat to human health through food poisoning and wound infection, causing an estimated 80,000 illnesses per year in the United States. As ocean temperatures continue to rise due to climate change, Vibrio infections are expected to sharply increase. Vibrio species employ highly complex systems to regulate the formation of robust biofilms that are relevant to host infection. My goal is to uncover the molecular mechanisms of critical proteins in these pathways, using a wide range of biochemical, biophysical, and microbiological approaches. This work will answer many basic science questions about biofilm regulatory mechanisms and will allow us to identify potential intervention points for drug targets.
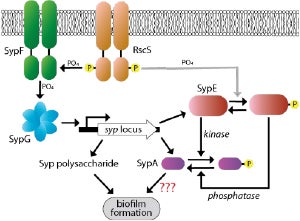 We focus on two main systems: the regulatory mechanism of the Syp pathway, and the link between quorum sensing and biofilm regulation. The first project is centered around the novel syp locus from NCVs. This locus encodes a complex network of proteins necessary for synthesizing and regulating the symbiosis polysaccharide (Syp) component of the biofilm matrix. My lab explores the structure and function of the regulatory proteins that play a central and critical role in controlling this pathway. The proteins in this pathway present many interesting and diverse scientific questions. The second project investigates the molecular communication between bacterial quorum sensing and biofilm formation pathways. Quorum sensing is crosstalk between bacteria through the exchange of small molecules that communicate cellular density. Once a bacterial population reaches a critical density, genes that control virulence traits such as biofilm formation are turned on by a signal transduction pathway. Compared to other bacteria, Vibrios have a complex quorum sensing pathway composed of several main signaling cascades, the Van/Lux phosphorelay. The lab is working on elucidating the molecular mechanisms of the proteins that link the quorum sensing pathway to biofilm formation.
We focus on two main systems: the regulatory mechanism of the Syp pathway, and the link between quorum sensing and biofilm regulation. The first project is centered around the novel syp locus from NCVs. This locus encodes a complex network of proteins necessary for synthesizing and regulating the symbiosis polysaccharide (Syp) component of the biofilm matrix. My lab explores the structure and function of the regulatory proteins that play a central and critical role in controlling this pathway. The proteins in this pathway present many interesting and diverse scientific questions. The second project investigates the molecular communication between bacterial quorum sensing and biofilm formation pathways. Quorum sensing is crosstalk between bacteria through the exchange of small molecules that communicate cellular density. Once a bacterial population reaches a critical density, genes that control virulence traits such as biofilm formation are turned on by a signal transduction pathway. Compared to other bacteria, Vibrios have a complex quorum sensing pathway composed of several main signaling cascades, the Van/Lux phosphorelay. The lab is working on elucidating the molecular mechanisms of the proteins that link the quorum sensing pathway to biofilm formation.
 The Milton lab uses a variety of biochemical, biophysical, and microbiology techniques to study the structures and function of proteins. These include X-ray crystallography, computational modeling, mass spectrometry, protein-protein and protein-nucleic acid assays, protein purification, and biofilm formation analysis.
The Milton lab uses a variety of biochemical, biophysical, and microbiology techniques to study the structures and function of proteins. These include X-ray crystallography, computational modeling, mass spectrometry, protein-protein and protein-nucleic acid assays, protein purification, and biofilm formation analysis.
If you would like more information about our research, potential collaborations, or joining the Milton lab, please contact Dr. Milton at miltonm19@ecu.edu
Selected Publications
Milton ME, Visick KL. (2023). Computational and cellular exploration of the protein-protein interaction between Vibrio fischeri STAS domain protein SypA and serine kinase SypE. Commun Integr Biol. 16(1):2203626 doi: 10.1080/19420889.2023.2203626.
Milton ME, Cavanagh J. (2023) The Biofilm Regulatory Network from Bacillus subtilis: A Structure-Function Analysis. J Mol Biol. 435(3):167923. doi: 10.1016/j.jmb.2022.167923.
View complete bibliography (NCBI) listing for Dr. Milton.
Recent Funding
National Institutes of Health
K22AI170662, 2023-2025
Title: “Regulatory mechanisms of novel host-relevant biofilm formation protein in non-Cholera Vibrio species.”
Wooten Family Initiative for Brain Health Research
Foundation Grant, 2023-2024, with Dr. Holly Ellis (co-PI)
Title: “Exploring the putative calbindin-D28k and caspase-3 interaction.”
